Click here for all the information you will need to produce your Science Fair project
Your browser does not support viewing this document. Click here to download the document.
Can you turn a liquid into a solid?
|
States of matter
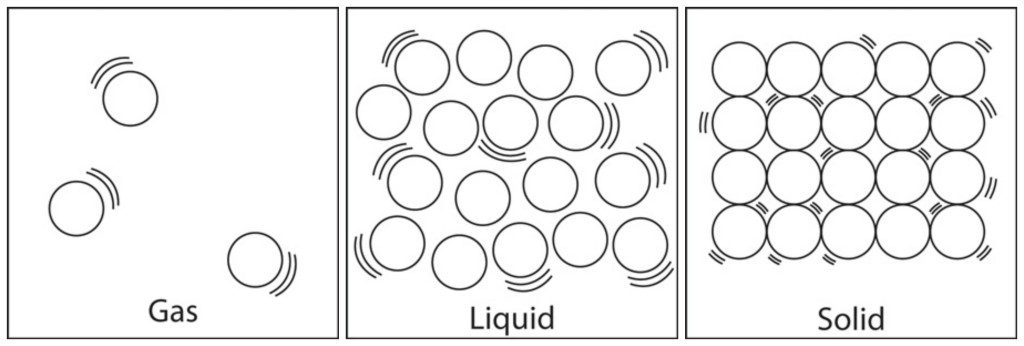
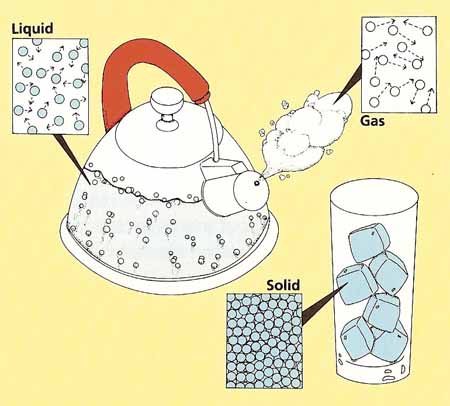
A material might be found in different states. The most familiar states are solid, liquid or gas.
The amount of energy the atoms or molecules of a material possess determines its state
of matter. A substance will exist as a particular state of matter in particular temperature and and normal air pressure water can be found as a liquid and iron is found as a solid. Increasing the temperature eventually changes solids to liquids (the iron will melt) and liquids to gas (the water will become vapour).
A material might be found in different states. The most familiar states are solid, liquid or gas.
The amount of energy the atoms or molecules of a material possess determines its state
of matter. A substance will exist as a particular state of matter in particular temperature and and normal air pressure water can be found as a liquid and iron is found as a solid. Increasing the temperature eventually changes solids to liquids (the iron will melt) and liquids to gas (the water will become vapour).
- Solids have atoms or molecules that are held together with rigid bonds. The atoms vibrate in place but they do not change position. This means that a solid holds its shape.
- Liquids have atoms or molecules that are held together with looser bonds that allow atoms or molecules to slide past each other. They stay close together and so occupy a constant volume of space. Thus a liquid can only be compressed a little bit, if at all. Due to the force. Liquids take on the shape of their container. The liquid state of matter is an intermediate phase between solid and gas. Like the particles of a solid, particles in a liquid are subject to intermolecular attraction; however, liquid particles have more space between them, so they are not fixed in position.
- Gases have particles that are not held together with bonds..Gases have three characteristic properties: (1) they are easy to compress, (2) they expand to fill their containers, and (3) they occupy far more space than the liquids or solids from which they form. An internal combustion engine provides a good example of the ease with which gases can be compressed.